Chances are you know someone who has a rare disease or know someone who cares for someone with a rare disease. Rare diseases are not actually rare, which I know sounds loco, but stay with me here. First, what IS a rare disease? In the United States we define it as any disease where 200,000 or fewer patients have it. In the EU they define it as a condition that affects less than 1 in 2000 people. Potatoh, potatoe. So yes, for any specific rare disease, there aren't a lot of peeps out there dealing with it. However, there are at least 6000 different rare diseases that we've identified, so while there may be just a few patients with a specific rare disease, there are MANY patients out there with SOME rare disease. US estimates are that we have 25-30 million people here with a rare disease. That's about 1 in ten!
Let's take a look at a couple of diseases at the far ends of rarity. First up is Ribose-5-phosphate isomerase deficiency or RPI deficiency for short. Patients with this condition have a problem with the enzyme or enzymes in that pathway and to be super scientific, bad shit builds up that shouldn't, accumulates in the brain and does bad stuff all over the body as well. So far, a grand total of THREE people with this condition have been found since it was discovered in 1999. Want to read about it? Here's a free paper that's pretty interesting. With only three patients, there is little research going on and zero efforts on treatments at this time.
Kidney cancer, and more specifically renal cell cancer, is also a rare disease. However, this disease is towards the other end of the spectrum, with about 75,000 new cases in the US this year. Still rare, but one that has lots of active research going on, drug development happening, foundations that support it, etc. When I first started working in the biotech industry, waaaaay back in the bronze age (we're talking pre-internet kids!), there was only one approved drug for kidney cancer and it's a bit of a crazy one. It would cure, flat out cure, about 7% of patients who took it, but it would actually kill about 2% because of the way it used your immune system to attack the cancer. These days we've learned more about how to harness the immune system to fight disease and there are now over 20 approved drugs for kidney cancer.
With so few patients, why would a drug company want to jump in and work on a treatment? I'm glad you asked young pendejos! Besides just wanting to help patients who don't have any treatments (honestly, not everyone in this industry just wants to make money) here are four major reasons.
1Orphan Drug Designation/Exclusivity

So, this is kind of a big benefit that the FDA grants drug developers. If you have a drug that you're developing for an "orphan" indication (rare disease), AND you have some data to back up that your drug might have benefit, then you can apply for and hopefully get "orphan drug designation." This gives you several benefits, but the biggest are certain tax credits related to R&D costs and also no fees when you file for approval (can be a couple of million buckaroos!). If your orphan drug is approved, then you get "orphan drug exclusivity" which gives you seven years on the market where no other drug with a similar way of working can compete with yours. Usually, if you have good patent coverage, that's better than the FDA exclusivity, but this rewards companies that develop drugs where patent coverage no longer exists but still go forward with the expensive work to develop the drug anyway. Just to not be US-centric, in the EU that exclusivity lasts 10 years.
2Sometimes, it's Easier to Develop a Drug in a Rare Disease
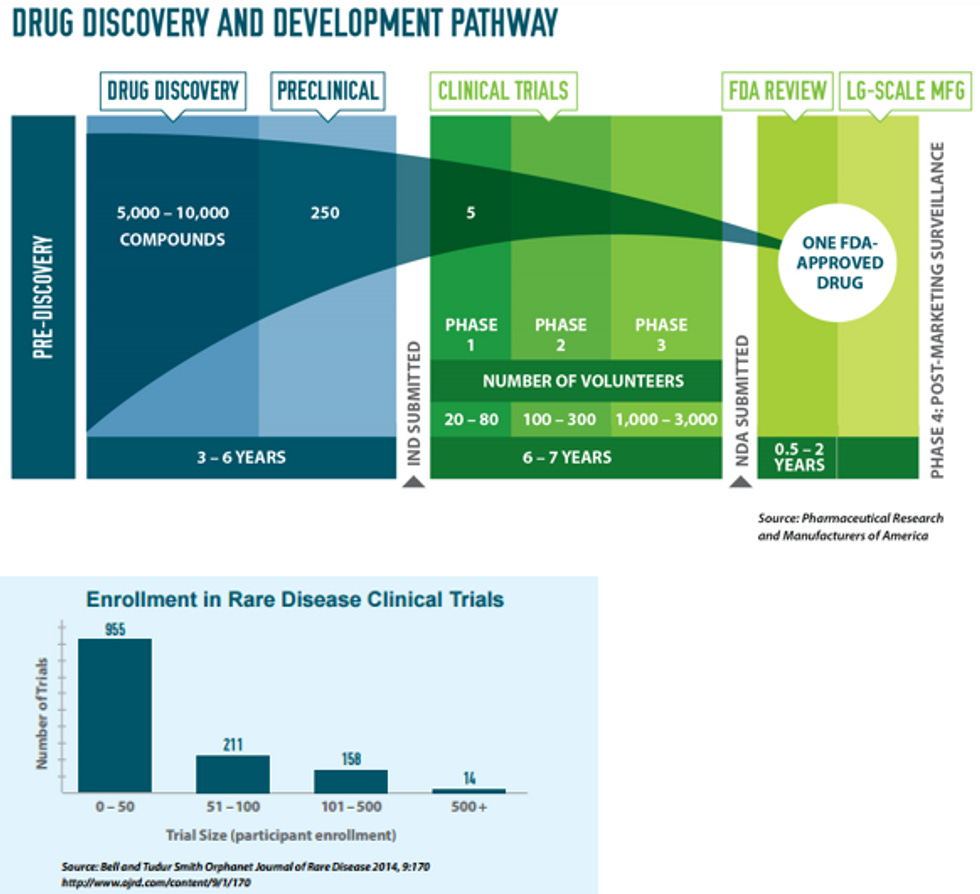
Unlike some high prevalence diseases like diabetes or Alzheimer's that can be caused by multiple factors, many rare diseases are genetic in nature and maybe it's pretty clear cut what's going on. For example, if you have a genetic mutation where one specific enzyme in your body is messed up, it might be as simple as creating a biotech version of that enzyme and giving you daily/weekly injections to replace the fucked up one with a shiny new one. Yeah, I'm way oversimplifying, but not THAT much. There are several approved drugs that work that way. Gene therapy is another approach looking at cures for these types of diseases.
3Sometimes It's Cheaper & Faster

Because rare diseases have so few patients, the regulatory agencies of the world that approve drugs usually let drug developers do smaller studies with less patients to get approved. Where a new hypertension drug might need to study 10,000 patients, I worked on a program where we got approval based on less than 100 patients in our development program. You still need to prove your drug works and is safe, but you can do it with smaller patient numbers. This can be much cheaper and sometimes faster. Sometimes faster, because while you need less patients, it can be VERY hard to find patients for the studies, because, and stay with me here, THEY ARE RARE DISEASES!
4You Can Charge a High Price for the Drug and Insurance Companies Will Cover it
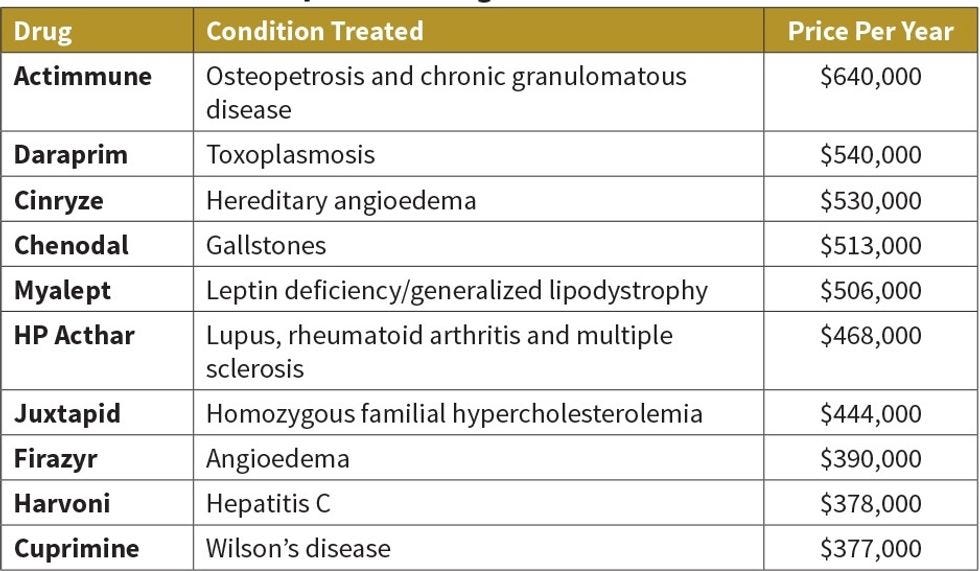
I know a drug that costs ~$400,000 annually for an average patient (weight-based dosing so more for bigger people and less for little people) but gets 100% coverage by the insurance companies and Medicare. Why no push back? One, it's life saving therapy. Two, it prevents lots of expensive medical procedures and delays (sometimes removes) the need for organ transplants. A life saver and a long-term cost saver to the companies paying for it. The other factor is that insurance companies don't care about the individual cost but the aggregate cost. If they have a million patients with a drug that costs a thousand bucks a year (cheap) that's a billion bones they have to pay out. If they have ten patients that cost half a million? Meh. Oh, and for the rare case where a patient can't get insurance to pay? This company gets them the drug for free. Honestly, we're not all terrible people that work in this industry!
Wonkette is ad-free and drug-company-free and we don't know why we didn't put a snarky little line at the end there, must be losing our touch.




I think that's the first time I've ever thought of drug companies in even a vaguely positive way. Thanks, Carlos!
Thank you for portraying us drug company worker bees in a flattering way, and educating us all about Rare Diseases in the process. I’ve been hearing about rare disease day all week (which, as a colleague pointed out, makes it sound like they are trying to give us all a rare disease), but this was the most interesting info I’ve heard.